Dr. Diane Haakonsen
Lunenfeld-Tanenbaum Research Institute
Switching off stress responses in health and disease
Cells in our bodies frequently encounter harmful situations such as heat, toxins, pathogens, including viruses and bacteria, and nutrient shortages. To cope, they have evolved essential cellular stress responses to protect themselves. But those responses become dysregulated in many diseases, including cancer and neurodegenerative disorders, and are often hijacked to fuel disease progression. Understanding how stress responses are regulated in both healthy and diseased cells is thus key to identifying new pathways and drug targets for future therapeutic strategies against these devastating diseases.
Our work has revealed that stress responses need to be actively turned off when their job is done, and that failure to do so can be highly detrimental. While much work has been done to understand how the appropriate stress responses are activated when needed, the brakes to silence them at the right time are not known and represent an interesting new avenue towards more effective treatments.
Our research investigates the mechanisms that turn off stress responses and how a failure to do so can lead to disease, including neurodegeneration. Our work takes a multidisciplinary approach combining genetic screens, cellular biology and biochemical and structural characterization.
Room 983, 600 University Avenue
Toronto, M5G 1X5
Publications: PubMed
Google Scholar: Diane Haakonsen
- 2024–present; Investigator, Lunenfeld-Tanenbaum Research Institute, Sinai Health, Toronto
- 2024–present; Assistant Professor, Department of Molecular Genetics, University of Toronto, Toronto
- Postdoctoral Fellow with Dr. Michael Rapé, University of California at Berkeley, Berkeley, CA, USA; 2016–2024
- PhD in Microbiology with Dr. Michael Laub, Massachusetts Institute of Technology, Cambridge, MA, USA; 2010–2016
- BSc, MSc in Life Sciences and Technologies, EPFL, Lausanne, Switzerland;
- 2025–present - Canada Research Chair in Cellular Stress Signalling
- 2018–2021 - Helen Hay Whitney Fellowship
- 2012–2015 - HHMI International Student Fellowship
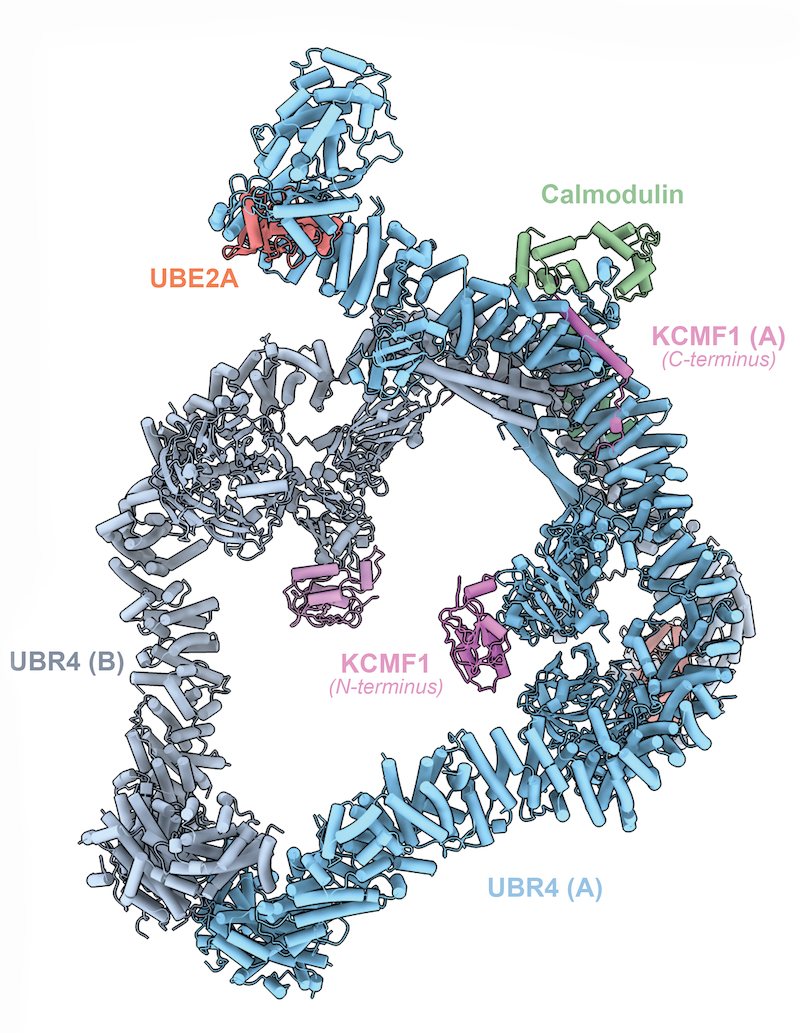
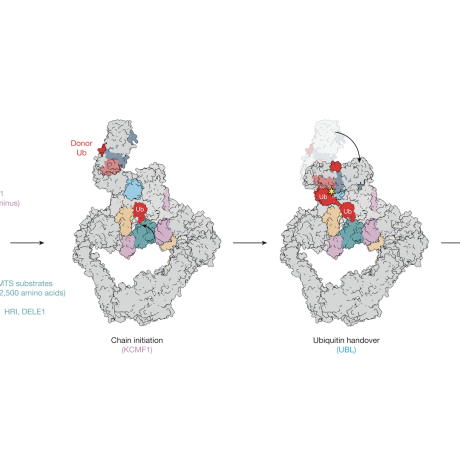
SIFI structure function analysis
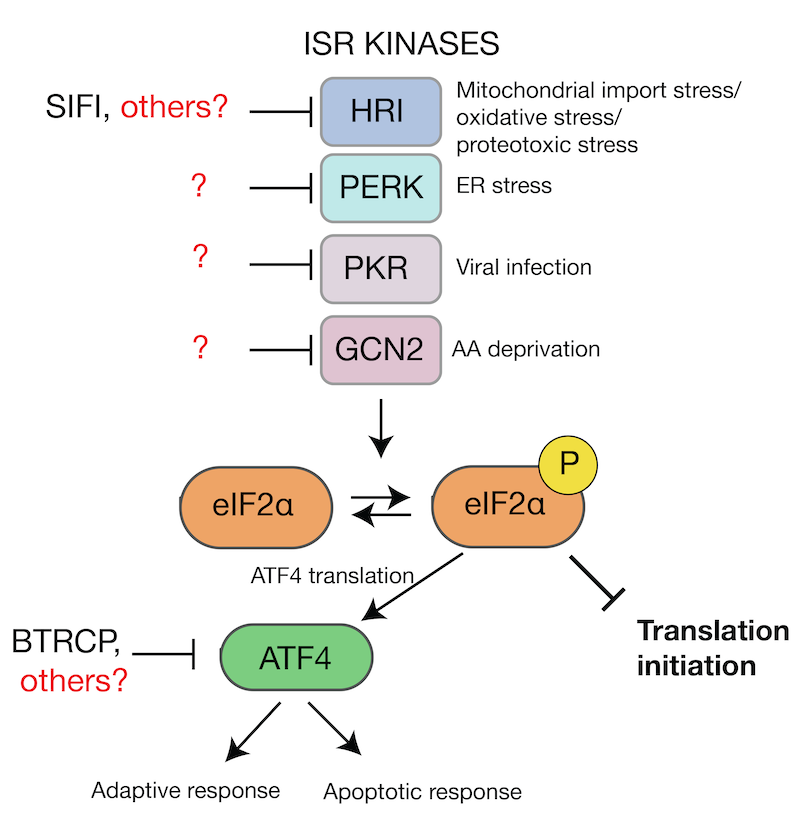
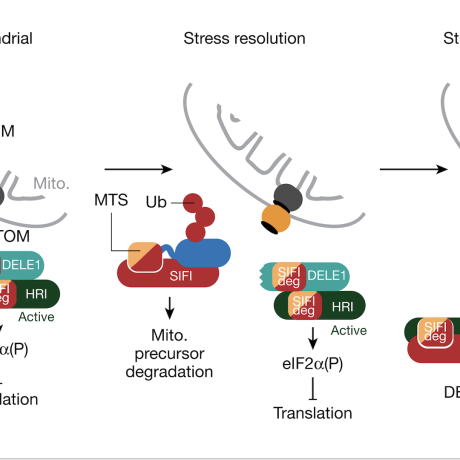
Mechanism of Integrated Stress Response silencing
ISR silencing in health and disease
Our work characterizing the molecular mechanism leading to silencing of the mitochondrial ISR has shown that it is required to restore cellular homeostasis. Interestingly, we found that silencing of the ISR dramatically improved survival in HEK293T cells experiencing mitochondrial stress, even when the mitochondrial import defect is still present (Haakonsen et al., Nature, 2024). These exciting findings suggest that in addition to the stress itself, chronic stress response activation may be an important contributor to neurodegenerative diseases caused by mitochondrial import defects. We are currently investigating this in neuronal and animal models.
We are always looking for motivated researchers to join our team.
Postdocs
Our research group is always interested in recruiting highly motivated postdoctoral fellows with a strong publication record in biochemistry, cell biology, structural biology (cryo-EM) or CRISPR screening. Please forward your CV, references and research interests to Diane Haakonsen ([email protected]).
Graduate students
Our research group is part of the Department of Molecular Genetics at the University of Toronto, which has a central admission committee and a rotation system. Graduate students interested in doing a PhD in the laboratory must first be accepted in the Molecular Genetics Graduate Student program.
Summer students
Summer students are exclusively selected from successful applicants to the Research Training Center (RTC) at the Lunenfeld-Tanenbaum Research Institute. Applications are available online and need to be filled by February 28th of each year.
Notable publications
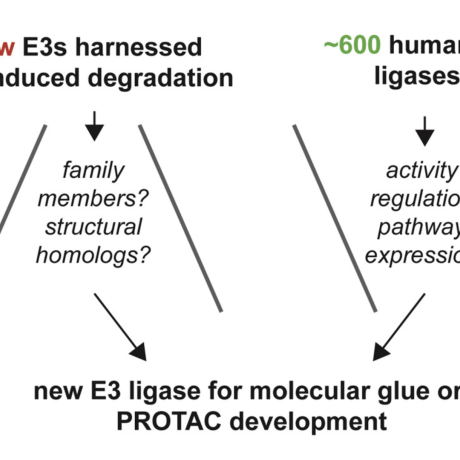
Cell Chemical Biology, 2021
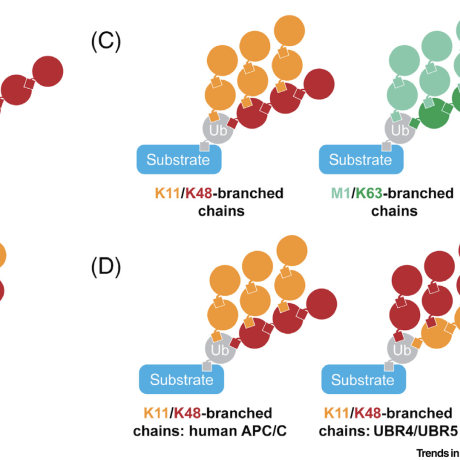
Trends in Cell Biology, 2019
Cell, 2017
Join our team
Visit our job board to see research positions.