Dr. Tatsuya Tsukahara
Lunenfeld-Tanenbaum Research Institute
In everyday life, animals including us humans constantly encounter diverse stimuli, or signals, that activate sensory systems such as the smell, sound, and scenery. To efficiently interpret these signals from the outside world in such dynamic environments and take appropriate actions at each moment, our brains adjust how they process these signals based on past experience. We recently discovered a new way the nervous system stays flexible. This process operates on long timescales (hours to days) in peripheral sensory neurons, which are the cells that connect us to the outside world, by converting past activity into changes in gene expression.
Building on this discovery, we use the mouse olfactory system, which is responsible for senses smell, to study studies how animals integrate signals from both the inside (hormonal and inflammatory signals) and outside of the body to fine-tune sensory responses. Most research has assumed that these sensory neurons simply pass information to the brain without changing, but we have shown that show they adapt based on experience. Our research will provide unique and critical insights into mechanisms for our remarkable ability to adapt to changes in external environment and our internal states, whose disruption is associated with various conditions such as autism spectrum disorders.
Email: [email protected]
Room 5-1004-3, 25 Orde Street
Toronto, M5T 3H7
Website: Tsukahara Lab
Publications: PubMed
Google Scholar: Tatsuya Tsukahara
ORCID: 0000-0002-6928-8562
LinkedIn: Tatsuya Tsukahara
Bluesky: @tsukaharalab.bsky.social
- 2023–present; Assistant Professor, Department of Molecular Genetics, University of Toronto, Toronto
- 2023–present; Investigator, Lunenfeld-Tanenbaum Research Institute, Sinai Health, Toronto
Former appointments
- 2012; Neurobiology course, Woods Hole Marine Biological Laboratories, Woods Hole, MA, USA
- 2010-2014; Assistant Professor (junior group leader in Hiroyuki Takeda’s laboratory), Department of Biological Sciences, Graduate School of Science, University of Tokyo, Tokyo, Japan
- Research fellowship with Dr. Bob Datta, Department of Neurobiology, Harvard Medical School, Boston, MA, USA; 2014-2023
- PhD, Biophysics and Biochemistry, University of Tokyo, Tokyo, Japan; 2007-2010
- MSc, Biophysics and Biochemistry, University of Tokyo, Tokyo, Japan; 2005-2007
- BS, Biology, University of Tokyo, Tokyo, Japan; 2001-2005
- 2021–2023 - Y. Eva Tan postdoctoral fellowship
- 2017–2018 - Louis Perry Jones postdoctoral fellowship
- 2015–2017 - Japan Society for the Promotion of Science postdoctoral fellowship for research abroad
- 2014–2015 - Astellas Foundation for Research on Metabolic Disorders postdoctoral fellowship for research abroad
- 2014–2015 - Alice and Joseph E. Brooks Foundation postdoctoral fellowship
- 2012 - William Randolph Hearst Educational Endowment
- 2012 - 28th Inoue Research Award for Young Scientists
- 2011 - GE & science prize for young life scientists, regional winner for Asia
- 2008–2010 - Predoctoral Fellowship of the Japan Society for the Promotion of Science (JSPS)
Long-term transcriptional adaptation in peripheral sensory neurons
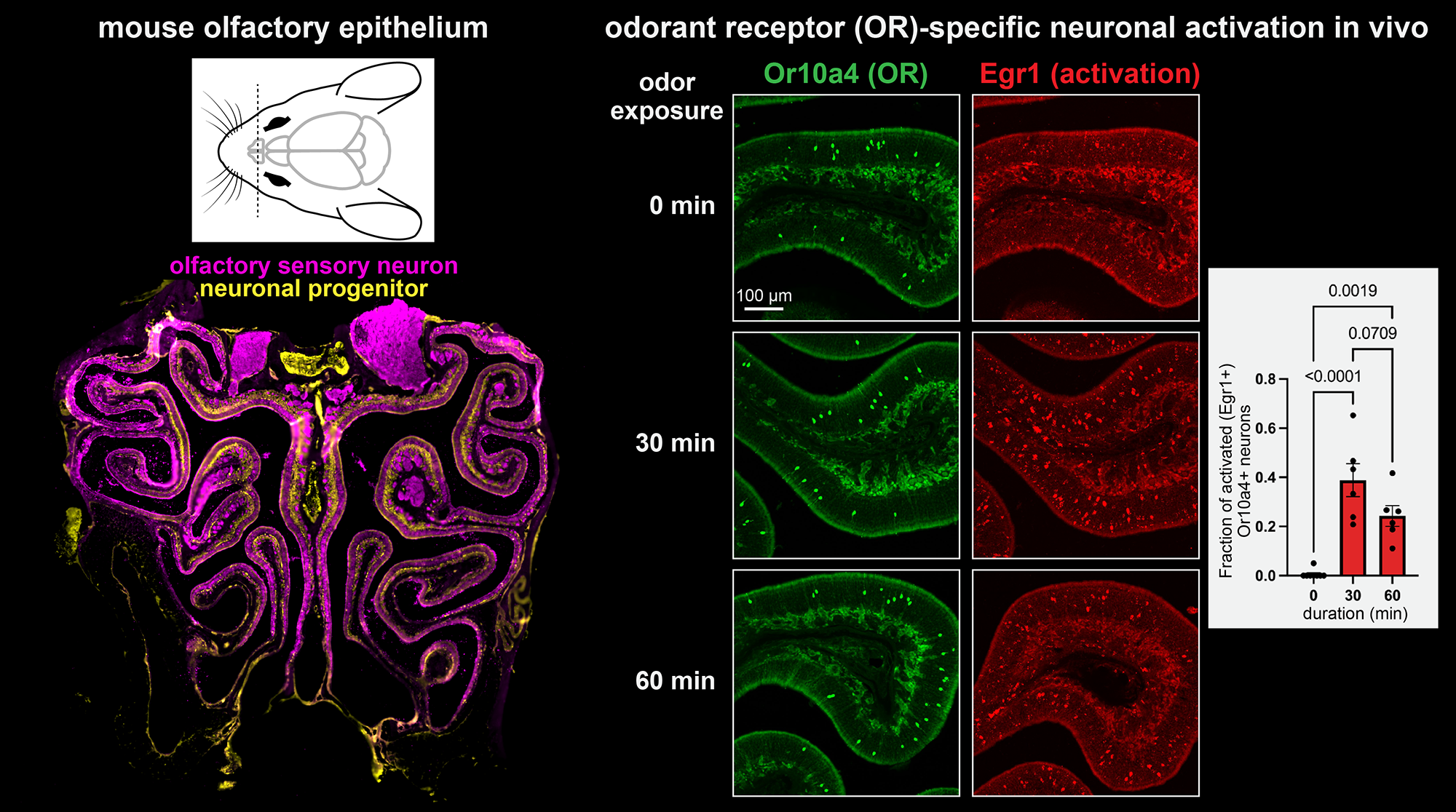
Neural activity induces large-scale gene expression changes. Although transcription and translation are important for long-term neuronal or synaptic plasticity, the underlying molecular mechanisms are still not fully understood. We recently discovered that olfactory sensory neurons in the mouse nose reconfigure transcriptomes and adapt the patterns of sensory responses to the activity history over hours to days. This study challenged traditional models that expect peripheral neurons to stably report sensory information to the brain irrespective of animal’s experience. In the brain, activity levels of neurons are determined by inputs from many neurons across different brain regions, challenging interpretations of their causes. Our use of the primary olfactory sensory neurons is advantageous because their activity levels are solely determined by odour molecules from the environment. This allows us to tightly control neural activity and to characterize the logic by which neurons convert activity changes into functional changes (neuronal excitability and synaptic outputs) via gene expression. Our lab aims to characterize 1) roles of immediate early gene transcription factors (e.g., AP-1 factors like Fos and Jun, Egr1) in adaptive changes in gene expression and sensory responses, 2) activity-dependent translational regulation of functional genes such as G-protein coupled receptor signalling, ion channels and calcium binding or signalling proteins, 3) roles of activity-dependent RNA transport and local translation at synaptic terminals in modulation of sensory inputs to the downstream neurons.
Inflammation-induced sensory neuron plasticity
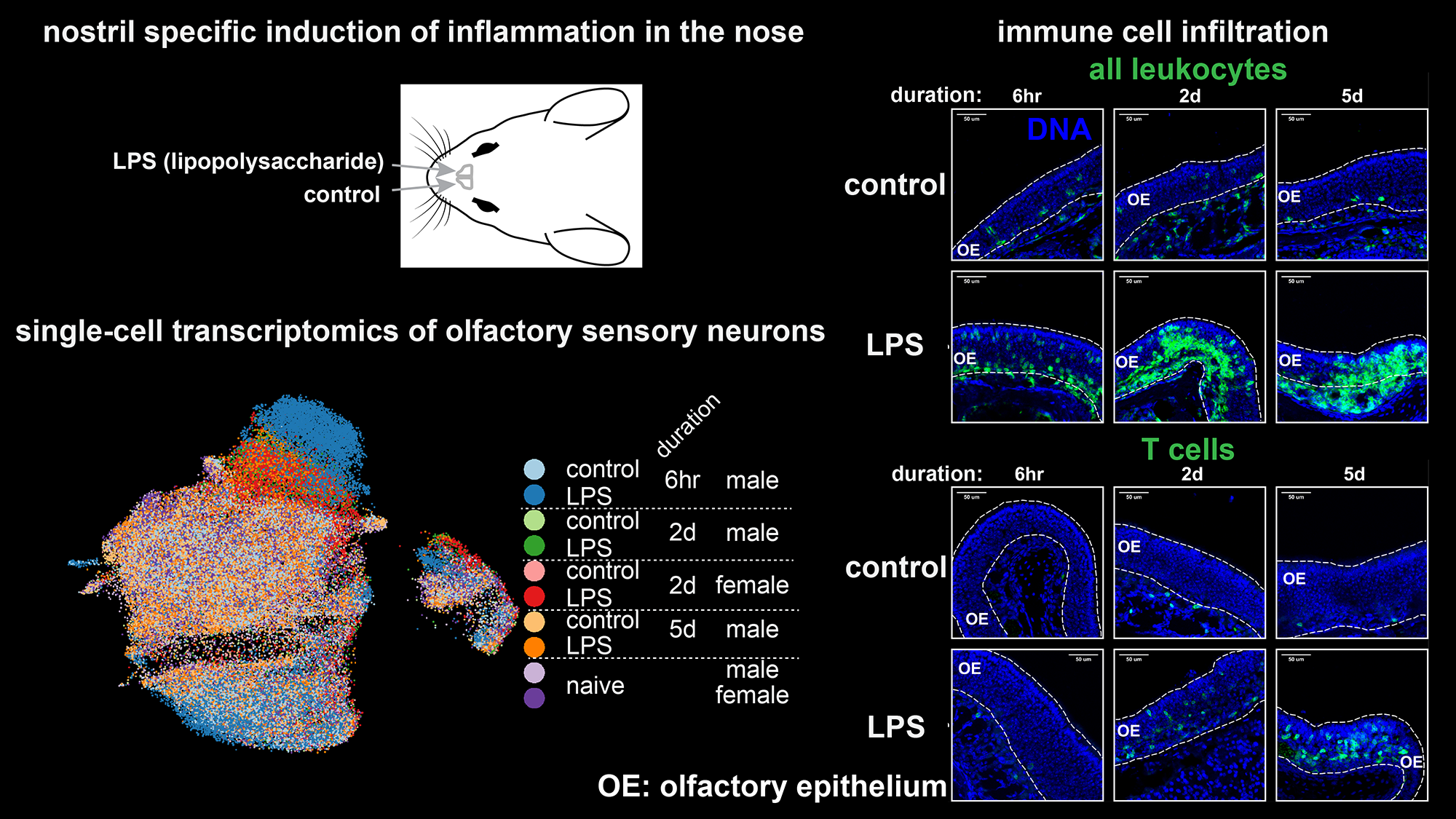
Inflammation is a body’s response to pathogens and cell death, characterized by recruitment of immune cells and release of cytokines, which can affect the nervous system functions. Cytokines can cause beneficial (sickness behaviour to fight off pathogens) or detrimental (neurodegeneration) effects via neuronal plasticity, and thus inflammation is considered as a key trigger for neurodegenerative diseases such as Alzheimer’s disease. Coincidentally, the impaired sense of smell is also an early marker of many neurodegenerative diseases (typically years earlier than cognitive decline). Furthermore, persistent inflammation in the olfactory epithelium in the nose where odourant molecules are detected is a major hallmark of long-COVID patients with olfactory impairment. Based on these circumstances, our lab aims to understand how inflammation impacts the olfactory system (especially olfactory sensory neurons in the nose), and how inflammation-induced plasticity in the nose contributes to the subsequent neurodegeneration in the brain. We use mouse models that mimic bacterial or viral infection and characterize the patterns of cytokine expression in the nose and their impacts in plasticity at neuronal (odour responses) and tissue level (neuronal survival and neurogenesis) levels, as well as odour-driven behaviours. We plan to expand our research to other conditions associated with inflammation and olfactory dysfunctions such as neurodegenerative diseases and obesity. In collaboration with Dr. Diane Haakonsen, we also study how mitochondria stress and cellular response to it reconfigure the neuronal states and lead to neurodegeneration by focusing on a mouse model of an accelerated neurodegenerative disease Deafness-Dystonia-Optic Neuropathy (DDON) caused by mutations in a key gene in mitochondrial protein transport pathway.
Hormonal regulation of sensory neuron plasticity and social behaviour
Social behaviour towards other individuals is central for our well-being and survival. Although social behaviour is a type of innate behaviour that can be executed without learning, its patterns can be drastically changed by going through life stages. For example, in mice, sexual experience transforms the behaviour towards pups from aggression to indifference to parenting in males, and indifference to parenting in females, at least partly through circulating sex hormones. Although prior studies have shown critical roles of a brain structure, the hypothalamus, in hormonal regulation of social behaviour, how these hormones (and other signals) modulate the sensory system is understudied. Nevertheless, growing evidence has shown that responses to social odours, even in peripheral sensory neurons, are modulated by life stages, which suggests an intriguing possibility that the nose is a unique organ to sense and integrate signals from the inside and outside of our body. Our lab studies how sexual experience (and becoming a parent) modulate the peripheral olfaction and how peripheral plasticity contributes to changes in social behaviour. In collaboration with Dr. Andrea Jurisicova, we also study how ovarian failure (menopause) reconfigure the peripheral olfaction by using mouse models of premature ovarian failure.
Nasally produced vasopressin (AVP)
Impaired social behaviour, such as difficulty in social communication and avoidance of eye contact, is commonly observed in people with autism spectrum disorders (ASD). An emerging target for alleviating social impairment in ASD is Arginine vasopressin (AVP), a peptide that control water homeostasis via systemic circulation and social behaviour as a neurotransmitter in the brain; AVP levels in blood and cerebrospinal fluid correlate with degree of social behaviour in humans and rodents (lower AVP results in less social behaviour), and intranasal delivery of AVP is shown to improve social behaviour in children with ASD. These results suggest that boosting AVP signalling may help alleviate behavioural alterations in ASD, even though AVP itself is not identified as an ASD risk gene and underlying mechanisms are still unknown. We recently discovered a rare population of olfactory sensory neurons in the mouse and human nose expressing AVP, which is among the first evidence of AVP-producing neurons outside of the brain to our knowledge. As olfactory sensory neurons detect odours and send the outputs directly to the olfactory bulb in the brain, it is possible that nasally-produce AVP plays pivotal roles in social behaviour and intranasal AVP augments this endogenous nose-to-brain pathway. To understand roles of these AVP-expressing neurons in social behaviour, our lab studies the downstream circuits in the brain and phenotypes of sensory neuron-specific knock-out in social behaviour, with a special focus on the interaction between parents and neonatal or juvenile mice.
We are always looking for motivated researchers to join our team.
Postdocs
Our research group is always interested in recruiting highly motivated postdoctoral fellows with a strong track record in neuroscience and/or systems biology. Please contact Tatsuya ([email protected]) with your CV, research interests, and three references if interested.
Graduate students
Our research group is part of the Department of Molecular Genetics at the Faculty of Medicine, University of Toronto, which has a central admission committee and a rotation system. Graduate students interested in doing a PhD in the laboratory must first be accepted in the graduate program in the Department of Molecular Genetics.
Undergraduate students
We are accepting undergraduate students through Research Opportunity Program (ROP) and several summer research programs (please visit our lab’s website for detailed information). Please contact Tatsuya with your CV, transcripts, and research interests if interested.
Notable publications
Cell, 2026
Cell, 2021
Science Advances, 2020
Nature Neuroscience, 2020
Join our team
Visit our job board to see research positions.